
But it is related to dipole moment and assertion is the explanation from bent angle so here reason is unable to give explanation of assertion. bond angle >SeO2 Lewis structure, molecular geometry, bond angle., Answered. Alternatively, the electron geometry of sulphur dioxide is in the shape of a trigonal. , SO2(Sulfur Dioxide) Lewis Structure, Hybridization, Molecular Geometry. S atom in both S O 2 and S O 3 is sp2 -hybridized. The molecular geometry of SO2 is curved, with a bond angle of 120. The extra repulsion of the lp vs double bonds accounts for reduction to 119. These areas also repel the bonds to the maximum distance apart, which is in this case 120 degrees. The molecule comprises one Sulfur Atom and two Oxygen atoms. In S O 2, we have 2 double bonds and one lone pair 3 electron regions so the default angle is 120 degrees. SO2 Molecular Geometry,Shape and Bond Angles (Sulfur Dioxide) Today in this video we are going to share a step-by-step procedure to determine the molecular geometry of SO2 molecules. The S O 2 molecule has a dipole moment, The starting point of 109.5 degrees is only applicable for 4 electron regions round the central atom. The bonding in S O 3 can be described as a combination of 3 resonance forms of 1 double and 2 single bonds. So the shape is based on a tetrahedral structure - but the extra repulsion of the non-bonding pair will decrease the O-S-O angles by a few degrees from the 'perfect' tetrahedral angle. There are 3 bonding sets of electrons and one non-bonding pair on the sulphur. 
SO2 (sulfur dioxide) has the same lewis structure, same resonance structures, and. Therefore the bond angle should be slightly less than 120 degrees. 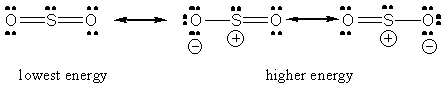
The ideal bond angle in a symmetrical trigonal planar molecule is 120° while the OSeO bond angle is approx. Be sure to identify the bond angles.The correct option is B If both assertion and reason are CORRECT, but reason is NOT THE CORRECT explanation of the assertion. All of the atoms are sp2-hybridized (+1 delocalized electron pair), which means the electron group geometry is trigonal planar (120 degrees in between bonds), and the molecular geometry is bent. It is due to the distortion present in the SeO 2 molecular shape and geometry that the OSeO bond angle decreases slightly from the ideal. Sketch the molecular shape of the compounds in the table below. Which of these three previous molecules will have all bond angles equal to each other? Explain completely.Ĥ. Aluminum bromide (Aluminium tribromide) AlBr3: Molecular Geometry - Hybridization. Subsequent replacements of H with Cl yield dichloromethane (CH2Cl2), trichlormethane (CHCl3) and tetrachlormethane (CCl4). As for the approximate bond angles the central atom with a bond angle. Examples of AX3 type molecules: BF 3, BCl 3, BBr 3, BI 3, HCHO. The central atom has sp 2 hybridization in AX 3 -type molecules. The ideal bond angle in AX 3 type molecule is 120. AX 3 -type molecules are ideally non-polar (Formaldehyde (HCHO) is an exception). Small bond angle distortions occur due to the lone electron pair on nitrogen. The molecular geometry or shape of AX 3 -type molecules is also trigonal planar. Chloromethane can be thought of as resulting from methane via the replacement of one Hydrogen atom with a Chlorine atom. The ideal bond angles in a trigonal pyramid are based on the tetrahedral electron pair geometry. What is the molecular geometry (shape) of the chloromethane molecule (CH3Cl)? Explain completely why NOT all of the bond angles in chloromethane are equal.ģ. What is the molecular geometry (shape) of the methane molecule (CH4)? Explain completely why all the bond angles in methane are equal.Ģ. Electron Pair Geometry So2 Together, the 10 non-bonding pairs and one single bond gives 22 valence. 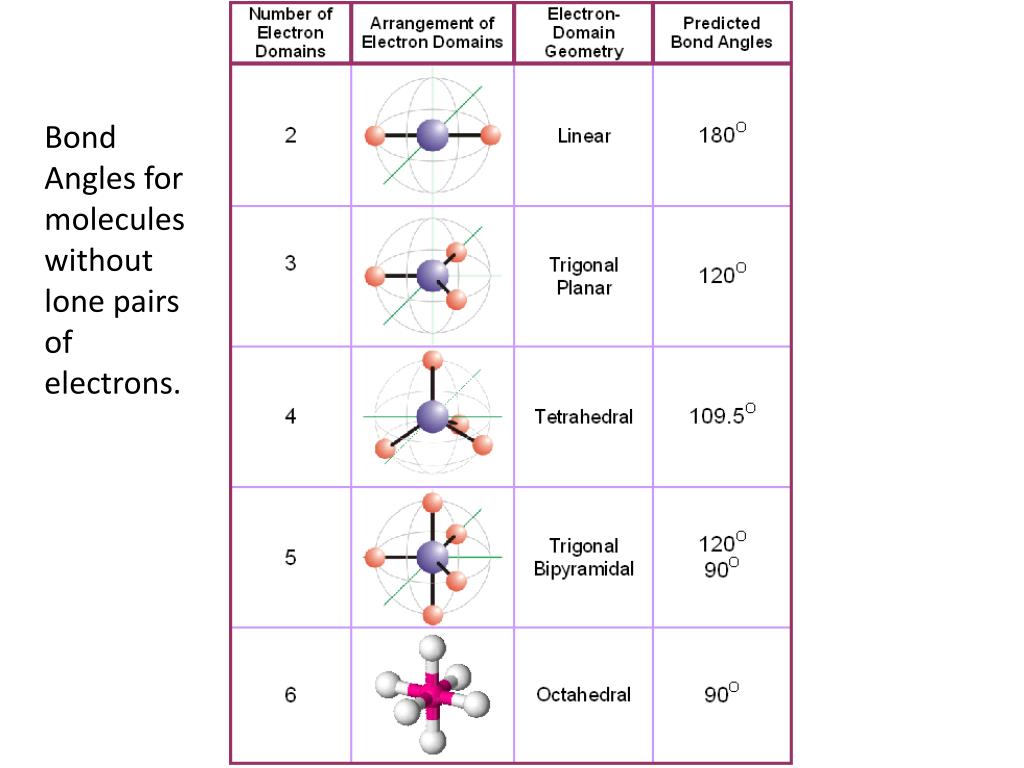
Lewis Structure, Molecular Geometry, Hybridization, Bond Angle and Shape. Part 2: Construct the three-dimensional geometry for the molecules listed in Table 2.ġ. The first step is to sketch the molecular geometry of the SO2 molecule, to. Part 1: Use your Periodic Table of Elements to determine the elemental symbol, group number and valence electrons for the elements listed in Table 1. In this experiment, you will predict the three-dimensional geometry of a series of neutral molecules using the VSEPR theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
